
Comentários
e sugestões para:
otavio@ingai.com.br
|
Haematological
profile and techno-patologies
Campanile
G., Di Palo R.,
d'Angelo A.*
Department
di Scienze Zootecniche, Università Federico II - Napoli
*Departement
di Strutture, Funzioni e Tecnologie Biologiche
|
Abstract
The
inter-relation between metabolic pattern and diet during the different
physiological phases in the buffalo are discussed herein, highlighting the
modification of the endocrine-metabolic pattern in the most critical phases of
productive life (early lactation, growth etc.).
In the buffalo the lesser milk yield is caused by a lower negative energetic
balance at early lactation compared to the cattle. This causes reduced fat
mobilization characterized by lower NEFA haematic levels.
Moreover, the buffalo adapts itself better than bovine to the greater energy:proteins
ratio and to the lower protein concentrations in the diet. Such a diet, over a
long period of time, does not lead to a reduction in the blood and milk urea
levels. High protein concentrations, on the other hand, at lactation-onset do
not show reduced azotemia (typical at early lactation) to meet proteic
requirements and does not cause an excessive rise in haematic ammonia.
Particular attention should be paid to mineral content during the dry milk
period as this could be one of the causes of uterine and vaginal prolapse. This
is due to alteration of uterine-vaginal muscular tone following the change in Ca:Mg
haematic ratio immediately post-partum.
Key words:
buffalo, nutrition, metabolic profile
Introduction
The
study of haematic constants help in identifying possible nutritional errors in
species of zootechnical inerest (Payne et al.1970) Moreover, they provide useful
indications on metabolic profile within the different physiological phases of
the subject. Zicarelli et al. have carried out studies with this objective on
Mediterrean buffaloes, bred in Italy, from 1980 onwards. Since then many other
researchers have contributed to the improvement of the buffalo diet troughout
the different productive stages (i-e,growth,dry milk period ,lactation ). The
aim of this work is to describe metabolic profile inter-relations during
different physiological phases, highlighting biological mechanisms which are at
the basis of productive activity in the subjects.
Energetic
Metabolism
Buffalo
lipomobilization, which increases NEFA (as observed in the bovine milk cow) (Bertoni
and Lombardelli, 1991; Chiesa et al., 1991) begins towards the end of gestation
(Campanile et al 1995). At early lactation, haematic concentrations of NEFA are
high, but never reach the levels found in the bovine milk cow, peaking at about
20 days post-partum (fig. 1). It than decreases (about 110 days post-partum )
returning to the haematic levels registered in the initial dry milk period phase.At
this point in time (about 110 days) an increase in insulin plasma levels is
noted. This indicates endocrine signal passage from the catabolic phase to the
anabolic (Campanile et al., 1995) on the lactation curve (fig. 1).
It
is already well-known that lactation start is characterised by a negative energy
balance which is more intense in the buffalo than in the bovine. We can
therefore confirm that buffaloes, even if fed a
well-balanced diet reach a "physiological" hyponutritive condition as
they are unable to ingest the quantity dry matter (D.M.) needed to meet requirements. The organism responds to this emergency by
raising GH levels and lowering those of T4 (functional hypothyroidism).
Figure
1. NEFA, B-OHB and insulin haematic levels at different days from calving.
NEFA,B-OHB
calving
Insulin
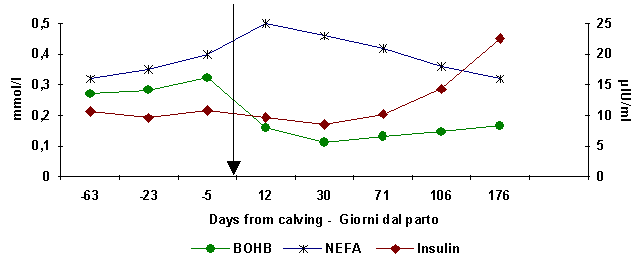
The
growth hormone reacts by reducing insulin activity, or at least sensitivity of
adipose tissue towards this hormone. This mechanism facilitates mobilization of
the adipose tissue and the muscular
proteins which provide energy and the necessary precursors in order to maintain
glucose haematic levels within values which allow the subject to carry out its
phisiological activity. Low glycemia values have been found in buffaloes which
intake less than 1020 Kcals per litre
of milk of NEl (net energy for lactation). In these buffaloes whose NEFA and
-OHB were not analysed, haematic levels
turned out to be inferior compared to what is deemed normal in these species (55
60 mg\dl) (Zicarelli et al., 1982) and
inversely correlated with the quantity of milk produced, but positively with
distance from calving (Zicarelli et al., 1982; Eltohamy et al., 1994). Other
studies have shown an increase of this haematic constant following a rise of T3
and T4 values in cold climates especially in winter (Campanile et al., 1994).
In
buffaloes, towards the end of the lactation curve, it is interesting to note a
possible increase of
-OHB (fig. 1) in a phase characterised
by lesser fat mobilization and a reduction in the quantity of short chain
fatty-acids present in milk fat. These events (noticed also by other autors) (Bertoni,
1994) could be explained by changing the group of a poor production buffaloes
which on Italian farms, is carried out after approximately 180 lactation days.
In this case, it has been noticed that the increased fill value of the diet and/or
to the change of the same which is administered to the less productive subjects.
There are two possible reasons for this: energy requirements are not sufficently
met in subjects which, at this phase, produce a milk particularly rich in fats
and/or stress derived from the re-estabilishement of group hierarchy. In same
cases from the 110th day of lactation, an increase of
-OHB can be observed upon increase of
insulin and trygliceride haematic levels. Bertoni and coll. (1994) found a
poorer use of
-OHB upon raising insulin values in
milk cows. Nevertheless,
-OHB increase has been noticed in
subjects whose daily requirements were more than sufficiently met and whose
diets contained high proteic and energetic levels (14.5% - 15%
CP/DM and 0.89 - 0.94
FMU/DM). The high proteic levels could have directed ruminal fermentation
towards greater production of butirric acid
which determines a greater quantity of circulating
-OHB not immediately used by the
organism.
Adding
supplements of protected fats both at lactation start and after
lactation peak (about 55 days
postpartum) causes fat mobilization increase in buffaloes (Di Palo et al., 1997)
and bovine (Sklan et al., 1994). In fact, higher NEFA and total cholesterol
plasma values are found, although the values of
-OHB are lower. Protected fat ingestion
should therefore stimulate production through an active mobilization of body
reserves which can guarantee the udder the precursors for the synthesis of the
milk components. The higher NEFA values in respect to the controls, do not
indicate energy defiency, but represent a more efficient use of
body reserves. The lower
-OHB values confirm this.
In fact, it means a compensated fat mobilization which positively
influences the energy balance (low
-OHB values), synthetic hepatic cell
activity (high cholesterol levels) and milk yield.
Total
cholesterol and HDL indirectly provide for evaluation of esogenous energy
availability together with hepatic functionality. When the energetic quota
consumed by the animals manages to meet at least part of its daily requirements
the increase of NEFA, following fat mobilization, does not cause lipid liver due
to trygliceride persistence in the hepatic cells.
In this case, the hepatic cells are not
capable of increasing the synthesis of cholesterol. In fact, cholesterol
(as is known) indicates good hepatic tissue lipoprotein
production which transport the triglycerides synthetized by the NEFA (Nefa
Acil CoA
-glicerophosphate
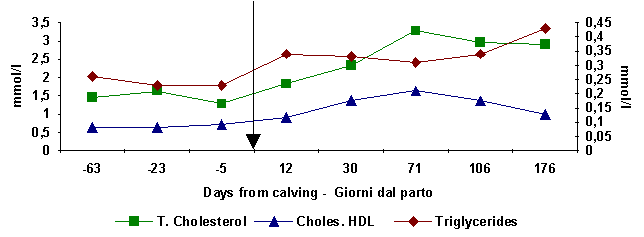
triglycerides). In buffaloes with a moderate negative energetic
balance, an increase in total cholesterol and HDL within 71 days of lactation is
observed (fig. 2). Then a lowering is found to coincide with the final stage of
the lactation curve and the beginning of the anabolic phase as soon as the
insulin (fig. 1) and triglyceride (fig. 2) haematic levels rise.
Particularly
high total cholesterol hematic levels are found in buffaloes bred in rather cold
climes (Campanile et al., 1994), in fact it is well known that catecholamine
which are stimulated by stress factors (these include metereological factors)
increase fat mobilization with rising
NEFA with the aim of producing
endogenous heat needed for thermoregulation.
Therefore,
lactation and low temperatures and/or high thermal ranges are resposible for the
energetic gap which probably causes fat mobilization with the introduction of
NEFA in circulation. In fact, total cholesterol is higher in subjects which move
more, but this does not cause energetic deficiency as the normal
-OHB values
prove.
Figure
2:
Total cholesterol, cholesterol HDL and triglycerides haematiclevels at
different days
from calving
(Total
Cholesterol)
calving - parto
(Choles.
HDL, Triglycerides)
Many
papers often highlight a good reletionship between total and HDL cholesterol and
milk yield. HDL cholesterol is less influenced by the dry matter (DM)
characteristic of the diet which means that it could represent a good genetic
index of the galactopoietic capacity of the buffalo (Di Palo et al., 1989).
Triglycerides
increase during the curve of lactation and show a positive correlation with the
milk fat levels (Zicarelli, 1988). In one study on 306 buffaloes with different
post-partum distances and fed 17 different diets, it emerged that triglycerides
represent a good index of meeting energy requirements, in
the early stages of lactation (1-31
days) while total cholesterol
and HDL in the intermediate phase (31-110 gg) and glycemia only after 110
days as it is positively correlated with the energy provided by the diet (Di
Palo et al., 1990). It is well know, in fact, that the NEFA which are released
at early lactation following intense fat mobilization, are used hepatically for
the syntesis of the triglycerides only if the balance between energy absorbed in
the diet and that emitted due to production is not especially deficit. Rising glycemia, on
the other hand, is obtained only during the anabolic phase of lactation when
energy intake is equal to, or superior to the energy release. Normal or high
values of hematic glucose are responsible for the lowering of GH and the
increase of the insulin:glucagon ratio which achieves a lesser or zero
efficiency in activating neoglucogenesis through other molecules (Aa etc).
Proteic
Metabolism .
In
buffalo cows, as well as in dairy cows, dietary protein characteristics and P/E
(protein:energy) ratio influence urea levels in blood and milk (Campanile et
al., 1996). This ratio (P/E) must be better evaluated considering that the
buffalo cow adapts itself to the lack of protein easier than the dairy cow (Bertoni
et al., 1993) It is necessary to remember that low values
of BU (blood urea) and MU (milk urea), compared to the cow, were reported only
when passing from 12% to a low 9% protein content diets (Campanile et al.,
1996). Proteic concentrations < 9%,used for a long time, did not determine
low blood urea values, but rather cause milk freezing point (MFP), especially if
the diet shows a high amount of fermentable energy (Campinile et al., 1996). It
would seem interesting, therefore, to evaluate the protein/energy ratio in order
to to obtain a correct diet. In the buffalo, such a ratio can be broader;
supplying diets high in proteic concentration determine in fact, less damaging
effects compared to the bovine milk cow. The buffalo, for example, makes better
use of nitrogen in the diet than the bovine, even if there is a carbohydrate
deficit (Langer, 1969) since buffalo ruminal behaviour is more favourable toward
the growth of micro-organisms which use non proteic nitrogen.
BU
and MU levels are positively influenced by the CP/NSC ratio also in the buffalo.
In fact, the greater availability of fermentable energy, which can be used by
ruminal microflora, could allow a better use of ammonia at ruminal level for
microbial protein synthesis. Therefore, a smaller quantity of urea could be
produced (Journet et al., 1975).
In
our experiment (Campanile et al., 1996) the length of the period during which a
diet with low protein concentration, as previously stated, was used, influenced urea levels in blood and milk. In fact, the urea
levels in blood and milk reached higher levels when a low-protein diet was given
for a fairly long period of time, whereas they were lower immediately following
a sharp reduction in the protein contribution of the diet. Prolonging the
absorption of low protein concentrations induced the subjects to maximise the
use of the fermentable organic matter for the production of bacterial proteins (Bertoni
et al., 1993); such a condition brought about the activation of a normal
aminoacidic breakdown which aided in keeping urea levels high. Following sharp
reduction in the protein content of the diet, a decline in circulating urea was
verified. This was possible when a fermentable energy supply was present, which,
in contributing to the increase of insulin levels, reduced or blocked the normal
aminoacidic breakdown, and, therefore, reduced the urea levels in circulation.
In
the dairy cow decreasing BU was registered exclusively in diets with low
nitrogen levels together with low energy levels (Ndibualonji et al., 1995). This,
on the other hand, is normal if the low nitrogen levels were associated with
high energetic levels. Smith (1969) reported that nitrogen deficiency in
ruminants which live in tropical areas reduces renal clearance of the urea,
increases the return to the rumen and decreases hematic levels. This, in turn,
would promote better recycling of the urea in the digestive tract and a better
proteic synthesis by the ruminal bacteria (Houpt, 1970).
Zicarelli
(1994) stated that the low proteic content of tropical forage, meets the daily
nutritive requirements of the buffalo vs the bovine more efficiently due to the
broader E/P ratio of the milk. Moreover, the more efficient recycling of the
urea in buffaloes explains their greater suitability and/or adaptability to
tropical and subtropical areas.
Azotemia
is influenced by days in milk and the diet, in fact, in buffaloes which intake
low proteic concentrations, it's progress reflects the modifications of dry
matter intake during lactation. The figure 3 shows that diets with 13.5% CP/DM
in subjects which yield more than 20 kg of ECM (equivalent
corrected milk), the azotemia presents at between the
11th and 32th day, low values which go back up between the 70th and 110th day
when dry matter intake increases. An accurate examination of the data suggest
that between the 11th and 70th day, dry matter intake is comprised of between 13
and 16 kg and therefore, the proteic requirements are not met. This leads to a
greater fat mobilization (NEFA increase) especially in the first days of
lactation (Fig. 3). It has also been noted that low azotemic values, in bovine
milk cows, cause a greater GH incretion which stimulates fat mobilization,
inhibits insulin activity and makes nitrogen derived of tissue so as to
guarantee the necessary quantity of energy and protein required for production
purpose (Ndibualonji et al., 1995). On farms which administer higher proteic
concentrations which we consider more suitable for meeting daily proteic
requirements in the period of lesser dry matter intake, especially in the more
productive subjects, lowering azotemia is not found (fig. 3). The greater
availability of keto acids, which derive from proteic catabolism, favours
glucose syntesis. Glycemia is therefore of higher in subjects with proteic
excess. High glucose values and low insulin values, guarantee the udder a
greater availability of glucose for lactose synthesis. This metabolic process,
used by subjects with proteic excess, contributes to reducing negative glucose
output, at the beginning of lactation. Spires and Clark (1979) have noted that
high levels of NH3 in steers, cause a poor use of the preceeding glucose, in
others words, a rapid glycogenolysis.
Interpretation
of the azotemia is carried out taking into consideration many nutritive aspects
and the integrity of renal parenchima. Our researchers generally evaluate
creatininemia (1.5 - 2.0 mg/dl) in order to exclude renal damage, which is
fairly fequent in this species.
In
the Mediterranean buffalo bred in Italy, excessive proteic diets are recorded,
especially in spring and early summer when pasture is abundant. Proteic excess
in bovine determines ruminal hyer NH3 with an increase of ammonia and urea
nitrogen levels in the blood. On the basis of our
experiences, with these diets the NH3 levels
have resulted lower than those reported by Jordan and coll. (1983) in
cattle; in these buffaloes an increase of the urea levels in blood and milk and
probably a greater elimination of the metabolite through urine occurred. Thus,
the increase of protein intake increases hepatic microsome metabolic activity
and this would favour the transformation of the ammonia of alimentary origin in
urea (Visek, 1984).
High
urea nitrogen levels have been observed by Zicarelli et
al. (1982) in buffaloes which consume more than 60 g of digestible proteins per
litre of milk. This however does not take place when 50% of the digestible
proteins came from wet brewers' grains which according to recent discoveries,
are not degraded at ruminal level by over 60% and are therefore endowed with a
good by-passability (Di Lella et al., 1995).
Infascelli
et al. (1997) found that in highly productive buffaloes (> 30 kg ECM/day),
the increase of UIP obtained from an extract of Aspergillus oryzae fermentation,
lead to greater milk yield and higher urea values.
Albuminemia
(2.6 - 3.9 g/dl) and total proteins (7-8 g/dl) are influenced by the proteic
levels in the diet only when there is excessive defficiency. These two constants
together with the A/G ratio (0.65-0.85) represent, on the other hand, a good
hepatic functionality index, as the positive correlations found between albumin
and the milk proteic level demonstrate. In buffaloes with extensive hepatic
necrosis, albumin levels lower than 2.3 g/dl were found. A reduction in the
ratio is also found in acute infections.
Figure
3:
Urea, Nefa, glucose and B-OHB haematic levels in buffaloes that were fed
diets characterized by 13.5% (1) or 15.5% (2) CP/DM at different days in milk
(NEFA)
(Urea)

Seric
Enzymes
The
cytoplasmatic enzyme, GGT (
-Glutamyltransferase), is the first to increase even in
conditions of slight hepatic sufferance. It is hightly correlated with SDH (Sorbitol
dehydrogenase) and OCT (Ornithine carbamoyl transferase) enzymes, those which
more specifically stabilize hepatic functionality, even if difficult to analyze.
Only in extremely serious cases, (for example with parasitic infestions of
hepatic type), it is possible to record AST (Aspartate aminotransferase) and
total bilirubin increases. It has been noted (Zicarelli et al., 1982) in
subjects analyzed in autumn and which feed at pasture between end-spring and
early autumn, higher values of Hb (Hemoglobin), AST and ALT (Alanine
aminotransferase). These enzymes seem to be present in ruminants, in greater
concentrations in the muscles and heart as opposed to the liver. Moreover, they
are localized at mitochondrial level for which they increase esclusively during
extensive hepatic necrosis. They represent, together with LDH (Lactate
dehydrogenase) and in particular CK (Creatine kinase), an index for the
evaluation of muscolar cells integrity.
The
determination of enzymatic activity is useful for ascertaining the validity of
the use of an acidified milk for wearing buffalo calves. The evolvement of
physiological conditions modifies the activity of the different apparatus in
this phase, and in particular one. The passage of the fetal and neonatal phase,
as well as tissue growth and change accompanied by turbulent synthesis which are
verified during this life-phase, could be responsible for the progressive
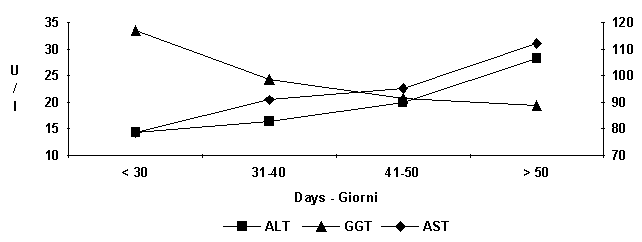
increase of the serum levels of ALT, AST and LDH (fig. 4) (Campanile et al.,
1993). On the other hand, the growth and therefore, the progressive and ever
more complete functionality of the organs, leads to greater activity of these
enzymes at tissuel level and consequently to their increase at serum level.
These findings togrther with normal albumin values and a progressive lowering of
GGT (fig. 4) during growth, have demonstrated an evolvement of synthesis
capacity and good functioning of the hepatic and renal parenchyma respectively (Campanile
et al., 1993).
The
progressively decreasing GGT values, together
with those of the
-globulins (according to some authors) (Tradati et al., 1982),
would be linked to the return within physiological limits of these two constants
which rise noticeably during the colostral phase, in as far as absorbed with the
colostrum.
The
verification of the A/G (albumin/globulin) ratio negatively correlated with GGT
( r = - 0.417; P<0.0001) also represents a valid testimony of good hepatic
functioning.
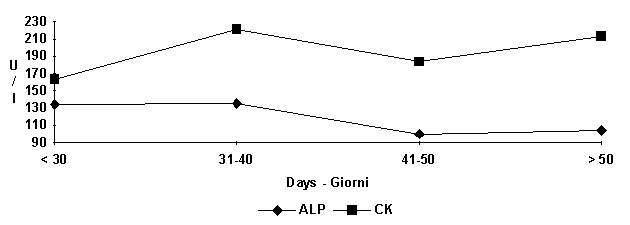
The
progress of CK (fig. 4) values is of particular interest. An increase on the
30th day of life is followed by progressive reduction; this progress could
represent the resultig increase of muscular tissue and functional gymnastics.
This
condition would cause, therefore, a greater permeability of the striated
musclolar cells and the limited liberation of CK within the circulatory system.
Higher values of ALP (Alkaline phosphatase) were found in the first 40 days of
life due to the more intense bone remodeling (fig. 4).
Climatic
variations also influence changing hematic constants. During the hottest weather
a lowering of the biosynthetic process of the organism as a consequent of the
lower basal metabolism is caused by the lower T3 and T4 hematic levels.
The
decreasing metabolism is accompained by lower hematic concentrations of ALT, AST,
ALP, LDH, GGT and PT (Di Palo et al., 1993). Moreover, thyroid hormones, and in
particular T4, determine an increase of mitochondria which swell due to the
increased membrane permeability, and therefore, a greater emission of these
enzymes in circulation.These hormones also stimulate proteic synthesis in
various organs.
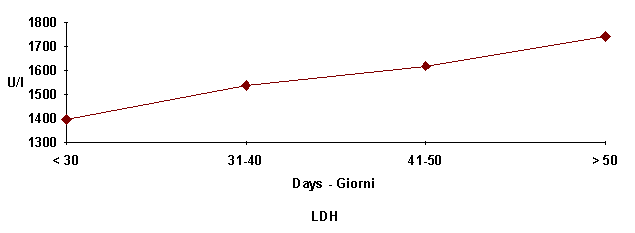
Figure
6:
Average seric enzymes pattern at different ages.
Mineral
Metabolism
The
hematic levels of the mineral elements and their relation help to evidence, not
only subclinical pathological alterations, but above all mineral deficiences. In
the dry period, even minimum, but long-term definciencies (easy to ascertain as
it is usual not to revise the diets of non-productive buffaloes) can cause
notable damage which is reflected in the state of health of the following
lactation. It has been noted that P (Phosphorus) deficiency during the dry milk
period represents one of the most frequent causes of vaginal and/or uterine
prolapse in this species (Campanile et al., 1989). Minerals salt supplements
should be added to the buffalo diet during the dry milk period to give 45 g of
Ca (calcium) and 45 g of P in order to keep the relation between these two
elements near to unity. Excess Ca even if minimum causes parathiroid
hipoactivity, which did not turn out to be ready, at end-gestetion and early
lactation, to
mobilize
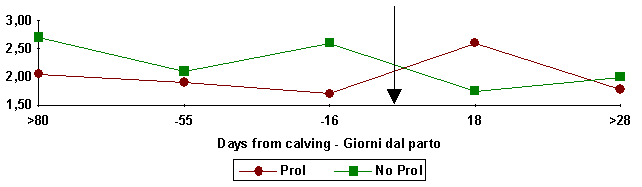
bone calcium. This determines excellent hematic Ca:Mg (calcium:magnesium) ratio
(fig. 5) alterations which is useful at maintaining membrane potentiality and
therefore permeability and excitability of the smooth muscular cells; this ratio
alteration favours utero-vaginal muscolar release, which provides atony and
therefore uterine prolapse (Campanile et al., 1989).
Figure
4:
Ca:Mg haematic ratio in buffaloes which have (Prol) vaginal prolapse or
not (No Prol)
In
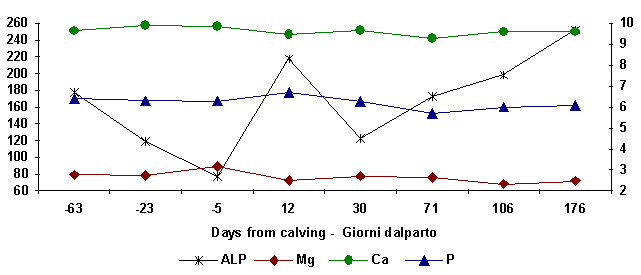
subjects whose diets were P deficient before calving, it was observed that
calcemia below 10 mg/dl decreased again to 8 mg/dl upon calving and phosphatemia
were normal (5-6 mg/dl) . Integration with P raises
calcemia speeding up
the glands which regulate homeostatis raising the values to 10mg/dl during the
dry milk period and to around 9mg/dl immediately after calving (Campanile et
al., 1989). In subjects whose dietary intake involves a Ca:P ratio nearing unity,
an increase of ALP (fig. 6) during the initial phases of lactation is proof of
speedy parathyroid activation (Campanile et al., 1995). In these subjects a
limited oscillation of Ca and Mg values was observed. High phosphorus hematic
values were noted (Zicarelli et al., 1982), together with higher Cu (copper)
levels, with subclinical metabolic acidosis in progress in diets characterized
by high quantities of silage and/or concentrates (> 59%). Metabolic acidosis
is responsible for both a greater incidence of uterine prolapse due to hypotonic
hydratation of the organ before calving (Zicarelli et al., 1982), and a greater
incidence of endometritis in the first 60 days from calving (Campanile et al.,
1991).
Circulatory
emissions of organic acids, which can be found during subclinical acidosis,
determines a lowering of the immune system resistence of the organism; which
leads to uterine colonization of the pathogens responsible for flogistic
phenomenon, and therefore, hypofertility. Moreover acidosis determines hepatic
sufferance proved by the higher values of GGT and LDH, and caused by the greater
effort by this organ in trasforming lactic acid into propionic acid (Campanile
et al., 1991).
Vitamins
Hematic
and milk deficiencies of B-carotene are found in buffaloes, probabily due to
quick transformation into vit.A (Zicarelli et al., 1986). Vitamins A and E
present the same trend of total cholesterol, in as far as they increase upon
increasing distance from calving to later reduce after 120 days from lactation.
Their average values are 597
g/l of vit.A and 175
g/l of the vit E (Zicarelli
et al., 1986).
Figure
5:
ALP, Ca, P and Mg haematic levels at different days from calving
(ALP, U/L)
(Ca, P, Mg, mg/dl)
Bibliography
Bertoni
G., Lombardelli L. 1991. Effetti dell'alimentazione sul quadro endocrino e
metabolico degli animali da latte. Atti IX Congr. ASPA, 1161-1189.
Bertoni
G., Amici A., Lombardelli R., Bartocci S., 1993. Variations of metabolic profile
and hormones in blood of buffaloes, cattle and sheep males fed the same diets.
Proc. of the Inter. Symp. "Prospect of buffalo production in the
Mediterranean and the Middle East", Cairo, Egypt, 9-12 November, 345-348.
Bertoni
G., Trevisi E., Bani P. 1994. Metabolic effects of two different lapses without
concentrate in early lactating dairy cows. Livestock Production Science, 39:
139-140.
Bertoni
G., 1994. Alcuni fattori che influenzano le condizioni endocrino-metaboliche
della specie bufalina. Agricoltura Ricerca, 153: 87-98.
Campanile
G., Di Palo R., Di Meo C., Boni R., 1989. Effetti dell'integrazione con P
durante l'asciutta sui livelli ematici di Ca P e Mg nella bufala. Atti XLII
Congr. S.I.S.Vet., Ottobre, 1:261-265.
Campanile
G., Bonavoglia C., Perrucci G.F., Di Palo R., 1991. Caratteristiche
ematochimiche in bufale appartenenti ad aziende con differente fertilità. Atti
Fe.Me.S.P.Rum., 1:93-97.
Campanile
G., Taccone W., Palladino M., Di Meo C., Di Palo R. 1993. Influenza dell'età e
del mese di prelievo su enzimi serici e quadro elettroforetico in vitelli
bufalini nella fase di allattamento. Nota I. Atti III Convegno della FeMeSPRum,
Teramo, 22-23 Ottobre, 21.1-21.9.
Campanile
G., Avallone L., d'Angelo A., Di Palo R., Di Meo C., 1994. Influence of the
season and of the number of days after calving on the pattern of thyroid
hormones in buffalo cows. IV World
Buffalo Congress, São Paulo, Brazil, June 1994, 3: 564-566.
Campanile,
G., Di Palo, R., Esposito, L., Boni, R. and Di Meo, C., 1995. Variazioni di
alcune costanti ematiche in bufale in lattazione. Atti XI Congr. Naz. A.S.P.A.,
Grado (GO), Italia, 19-22 June, 77-78.
Campanile
G., De Filippo C., Di Palo R., Taccone W., Zicarelli L., 1996. Influence of
dietary protein on urea levels in blood and milk of buffalo cows. VIIIth
Internat. Symp. of Vet. Lab. Diagn., 48 (Abstract).
Chiesa F., Gaiani R.,
Formigoni A., Accorsi P.A. 1991. Modificazioni del quadro endocrino e metabolico
in bovine da latte ad elevata potenzialità produttiva durante l'asciutta e la
lattazione. Arch. Vet. It., 42, 4: 157-179
Di
Lella T., Infascelli F., Cutrignelli M.I., 1995. Rumenn degradability and
proteic value of feeds utilized by buffaloes. Bubalus bubalis, 1: 38-48.
Di
Palo R., Di Meo C., Ferrari G., Zicarelli L. 1989. Interrelazioni tra alcune
costanti ematochimiche e produzione quanti-qualitativa di latte nella bufala.
Riv. Zoot. Nutr. Anim., 15: 489.
Di
Palo R., Campanile G., Boni R., Di Meo C., Zicarelli L. 1990. Interrelazioni tra
caratteristiche della sostanza secca della dieta, produzione lattea e alcune
costanti ematochimiche nella bufala. Atti XLIV Conv. Naz. SISVet., 1501-1505.
Di
Palo R., Campanile G., Palladino M., Spadetta M., Esposito L. 1993. Influenza
dell'età e del mese di prelievo su alcune costanti ematochimiche in vitelli
bufalini nella fase di allattamento. Nota II.
Nota I. Atti III Convegno della FeMeSPRum, Teramo, 22-23 Ottobre,
22.1-22.7.
Di
Palo R., De Filippo C., Princigalli D., Campanile G., Zicarelli L., 1997. Effect
of dietary supplementation with cryo-crystallized fatty acids on milk production
and metabolic profile in buffalo cows. Proceeding
V World Buffalo Congress, Caserta, Italy, October 1997, 372-377.
Eltohamy
M.M., Ahmed W.M., Abdon A.S.S., 1994. Ovarian activity in relation to some blood
composition. IV World Buffalo Congress, São Paulo, Brazil, June 1994, 3:
612-614.
Houpt
R., 1970. Trasfer of urea and ammonia to the rumen. In: Physiology of Digestion
and Metabolism in the Ruminant (AT Phillipson ed), Oriel Press Limited, New
Castle Upon Tyne, Royaume-Uni, 119-131.
Infascelli
F., Galiero G., Costa M., Sarubbi F., Di Lella T., 1987. Effect of Aspergillus
Oryzae on milk yield in buffalo cows. Proceeding V World Buffalo Congress,
Caserta, Italy, October 1997, 245-248.
Jordan
E.R., Chapman T.E., Holtan D.W., Swanson L.V., 1983. Relationship of dietary
crude protein to composition of uterine secretions and blood in high-producing
postpartum dairy cows. J. Dairy Sci. 66: 1854-1862.
Journet
M., Verite R., Vignon B. 1975. L'azote non protéique du lait: facteurs de
variation. Le Lait, Mars-Avril, n. 543-544: 212-221.
Langer
P.N., Sidhu G.S., Bhatia J.S., 1969. A study of the microbial population in the
rumen of buffalo (Bos bubalus) and zebu (Bos indicus) on a feeding regimen
deficient in carbohydrates. Indian J. Vet.Sci. 38: 333-336.
Ndibualonji B.B.,
Dehareng D., Godeau J.M. 1995. La sous-alimentation énergétique et/ou azotée
chez la vache tarie. Effect sur les acides gras volatils du rumen, quelques métabolites
et hormones du plasma et l'excrétion urinaire de la 3-methylhistidine. Reprod.
Nutr. Dev. 35: 137-154.
Payne J.M., Dew S.M.,
Manston R., Faulks M., 1970. The use of a metabolic profile test in dairy herds.
Vet. Rec., 87: 150-157.
Sklan
D., Kaim M., Moallen U., Folman Y., 1994. Effect of dietary calcium soaps on
milk yield, body weight, reproductive hormones, and fertility in first parity
and older cows. J.Dairy Sci., 77: 1652-1660.
Smith,
R. H., 1969. Reviews of the progress of dairy science. J. Dairy Res., 36:
313-331.
Spires H.H., Clark J.H. 1979.
Effect of intraruminal urea administration on glucose metabolism in dairy steers.
J. Nutr. 109: 1438-1445.
Tradati
F., Martinelli I., Besana V., Greppi G., Ferro E. 1982. Attività serica della
LDH, CPK e ALP in bovine e vitelli nel periodo perinatale (Note I e II). La
Clinica Veterinaria, 105: 185-191.
Visek
W.J., 1984. Ammonia: its effect on biological system, metabolic hormone, and
reproduction. J.Dairy Sci., 67:481-498.
Zicarelli L.,
Intrieri F., de Franciscis G., Squillaciotti S., 1982. Il profilo metabolico
nella bufala gravida in relazione al regime alimentare adottato: indagine in
allevamenti con diversa incidenza di prolasso vaginale. Atti II Conv.Int.
Sull'allevamento bufalino nel mondo, 1:262-288.
Zicarelli L., Avallone L.,
Salvatore M., Pizzuti G.P. 1986. Comportamento di alcune costanti ematiche nella
bufala in gravidanza e in lattazione. Atti XL Conv. SISVet., 569-572.
Zicarelli L. 1988. Il profilo metabolico
nella bufala. Inf. Zootecnico, 17:103-105.
Zicarelli
L., 1995. Management in different enviromental conditions. Buffalo J., sup. 2:
17-38.








